Receptors
Olfactory receptors are located in the olfactory epithelial tissue, in the upper part of the nasal cavity.
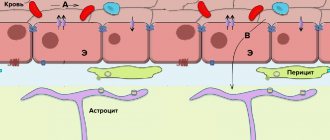
Rice. 1. Olfactory epithelium.
Each receptor cell has hairs facing the nasal cavity. The hairs are covered with a thin layer of mucus, since the smell and taste receptors are chemoreceptors that perceive the effects only of substances dissolved in water.
TOP 2 articles that are read along with this
- 1. Taste
- 2. Biology (8th grade): Visual analyzer
When molecules of odorous substances enter the nasal cavity with inhaled air, these molecules interact with the proteins of the olfactory hairs, and a nerve impulse occurs.
Perception of olfactory information
| External images | |
Olfactory cells
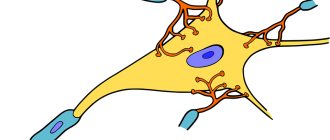
Olfactory receptors (olfactory cells) are bipolar neurons with one non-branching dendrite. It passes between the basal cells and ends in a small swelling - the olfactory club
.
Up to 20 long cilia emerge from it, which represent the sensory surface of the olfactory cell. They are usually immersed in a layer of mucus covering the epithelium and form a dense matrix with it. The olfactory cell has a dual function: perception of a stimulus and transmission of a nerve impulse to the brain, therefore it is a neurosensory cell (sensory neuron). Axons that transmit signals to the central nervous system are collected in bundles - olfactory filaments
. Olfactory neurons are capable of replacement by dividing basal cells[20][21].
The mucus in which the olfactory cilia lie contains a large number of small (20 kDa) proteins that are secreted by the nasal glands and are found in the mucus that covers not only the olfactory epithelium, but also the purely respiratory one. These proteins probably bind very non-selectively to molecules of odorants (odorants) and ensure their interaction with receptor cells[20].
Olfactory cilia do not differ in ultrastructure from other cilia and contain a regular stationary axoneme. Olfactory cilia are very long and thin: with a length of 5 to 250 microns, they reach only 100-250 in diameter. They are collected in bundles of 5-40 and emerge from the club of the olfactory cell, increasing its sensory surface. Receptor proteins are located on the surface of the cilia. Each gene from a family of genes encoding such proteins encodes a specific type of them, and on the cilia of one olfactory cell there are only one type of olfactory proteins; not all genes of this family, however, can be expressed (for example, about 40% of these genes are expressed in humans). For a long time, it remained unclear whether the cilium responds to many types of odorants or only to one[11]. Now, however, it has been established that olfactory cells of one type are specific to a specific narrow class of chemical compounds, since they recognize special structural motifs in them [14] [21].
Regardless of the specificity, the sensitivity of olfactory cells is very high: they are capable of detecting substances in concentrations from 10-4 M to 10-13 M. With a cold, sensitivity decreases due to the fact that the cilia are immersed in too thick a layer of mucus [11].
In addition to the olfactory cells associated with the olfactory nerve, the nasal mucosa also contains free endings of the trigeminal nerve; they are able to react to some aggressive odors, for example, acidic or ammonia fumes[21].
Carrying out the signal
The delivery of the olfactory stimulus begins as follows. The odorant binds to a receptor in the membrane of the olfactory cell. The olfactory receptor is a G protein coupled receptor and, like all GPCRs, contains 7 domains. Unlike other receptors of the GPCR superfamily, olfactory receptors are characterized by a large amino acid diversity in transmembrane domains[en] 3, 4 and, especially, 5. In addition, olfactory receptors differ from other GPCRs in less specificity: they have, to varying degrees, affinity to a number of stereochemically similar odorants. However, small changes in the chemical structure of the odorant may correspond to a change in the set of stimulated receptors and a change in subjective perception. Thus, replacing the hydroxyl group of octanol with a carboxyl group leads to a significant change in olfactory perception: instead of an odor reminiscent of an orange, the smell of rancidity and sweat is felt. In addition, the number of receptors stimulated and subjective perception may depend on the concentration of the odorant. For example, in low concentrations, indole has a pleasant floral aroma, and in high concentrations it has a disgusting putrefactive aroma[22].
The binding of the odorant to the receptor activates the Gs protein, which activates the enzyme adenylate cyclase, as a result of which GTP breaks down into phosphate and HDP. Adenylate cyclase converts ATP into cAMP, which binds to a cyclonucleotide-dependent cation channel in the membrane and opens the flow of Na+ and Ca2+ ions into the olfactory cell, thereby triggering an action potential in it, which is then transmitted to afferent neurons [21]. Sometimes olfactory receptors, however, activate not adenylate cyclase, but phospholipase, and the secondary messenger is not cAMP, but inositol triphosphate and diacylglycerol. In addition, it is possible that NO synthase is formed in olfactory cells due to calcium activation, which leads to the formation of cGMP [23].
Cyclonucleotide-gated channels have six hydrophobic segments and are structurally similar to voltage-gated ion channels. The difference is that cyclonucleotide-dependent channels have a large C-terminal cytoplasmic domain that binds to second messengers. There are 2400 channels/μm² on the cilia (on the olfactory club and dendrite there are only 6 channels/μm². In the absence of calcium, cyclonucleotide-dependent channels are permeable to all monovalent cations: Na+ > K+ > Li+ > Rb+ > Cs+. When exposed to an odorant, ion currents through cyclonucleotide- dependent channels change, leading to depolarization of the cell membrane and triggering an action potential[24].
Olfactory cells of the same type transmit their signals to the same glomerulus[en] of the olfactory bulb, and the spatial organization of the latter topographically repeats the location of the receptors on the surface of the olfactory concha[14]. It is worth noting that one olfactory receptor can be excited by one molecule of an odorant[25].
In 2004, Linda Buck and Richard Axel received the Nobel Prize in Physiology or Medicine for their research on mammalian olfactory receptors[26]; It was they who established the chemical nature of olfactory receptor proteins, estimated the number of genes in the mammalian genome encoding these proteins, and substantiated the rules according to which one olfactory cell expresses one type of olfactory receptor proteins, and one is responsible for processing signals from all olfactory cells of the same type the same glomerulus of the olfactory bulb[27][28].
Sensory adaptation
Interestingly, the cyclonucleotide-dependent channels of olfactory cilia do not become desensitized, that is, they do not lose sensitivity upon repeated presentation of the odorant. However, adaptation nevertheless occurs in olfactory cells. This is likely due to the entry of Ca2+ ions into the cell, which either directly or through activation of calmodulin lead to the closure of ion channels and, in addition, desensitize GPCRs[29].
In addition, the response to an olfactory stimulus is gradual, that is, a greater concentration of odorant corresponds to a greater response. This is due to the fact that cAMP increases or decreases the number of open cyclonucleotide-gated channels. To effectively distinguish signals in real time, a fast response is necessary. It has been shown that the peak of cAMP formation occurs 40-75 after exposure to an odorant and drops to zero after 100-500 ms. The G-protein cascade amplifies the signal, allowing one odorant pulse to activate multiple channels. However, the kinetics of the channels is quite slow, and the open state can lag behind the cAMP pulse by several milliseconds. With prolonged activation of GPCR odorants, cAMP pulses ensure the maintenance of cyclonucleotide-dependent channels in a constantly open state [29].
Olfactory bulb
The olfactory bulb is located above the nasal cavity, on the ethmoid bone. This is a paired organ that receives a signal from the optic nerve and performs primary processing of the received information.
Rice. 3. Olfactory bulb.
The olfactory bulb belongs to the forebrain and is considered a peripheral part of the olfactory brain, i.e., the brain structures involved in smell.
OLfactory ANALYZER
Olfactory analyzer
- a morphophysiological system that provides the perception of various odorous substances (smells), analysis and processing of the afferent excitations that arise.
The olfactory analyzer belongs to the chemical analyzers of distant action. Odorous substances reach the olfactory receptor cells located in the upper parts of the nose when they are inhaled through the nose or mouth and spread into the nasal cavity through the nasal pharynx and choanae. The act of eating is also accompanied by both olfactory (see Smell) and gustatory sensations (see Taste).
Rice. 1. Schematic representation of the peripheral section of the olfactory analyzer: 1 - olfactory cells of the nasal mucosa; 2 - axons of olfactory cells forming a nerve; 3 - cribriform plate of the ethmoid bone; 4 - olfactory glomeruli of the bulb; 5 - layer of fibers of the olfactory nerve; 6 - layer of olfactory glomeruli; 7 - reticular layer; 8 - mitral cells forming the mitral layer; 9 - granular cells of the olfactory bulb; 10 — cells with plumes; 11 - collaterals of the axial cylinders of the mitral cells in the olfactory bulb; 12 - collaterals of the axial cylinders of the mitral cells of the olfactory tract; 13 - pyramidal cells of the olfactory tract; 14 - centrifugal fiber: a - olfactory bulb; b - olfactory tract; c - olfactory triangle.
Peripheral part
The olfactory analyzer is represented by receptor cells located in the olfactory epithelium of the nasal mucosa, and the olfactory nerve formed by their axons (Fig. 1).
Receptor cells in humans are approx. 10 million, for a rabbit approx. 100 million, in dogs St. 200 million. Receptor cells of the Olfactory Analyzer are functionally and embryogenesis-wise similar to central neurons, but unlike them, they are capable of regeneration. There is evidence that throughout the life of the organism they are constantly renewed, developing from blastocytes of the basal layer of the olfactory epithelium. Replacement of receptor cells on the periphery leads to the renewal of synaptic connections of their axons without disrupting the functioning of the axon; the mechanism of this phenomenon has not yet been studied. Rice.
2. Relative sizes of the olfactory bulb (indicated by arrows) in humans and animals: 1 - cats; 2 - foxes; 3 - chimpanzees; 4 people; (1, 2 - macrosmatics; 3,4 - microsmatics). Olfactory nerve fibers
(see) end in the olfactory bulb, where they form specialized synaptic contacts—glomeruli. In these glomeruli, sensory information is transmitted to secondary neurons. The degree of convergence of impulses from receptor cells is very high: in each glomerulus, several dozen secondary neurons of the olfactory bulb receive afferentations from tens of thousands of receptor cells. Elements of spatial correspondence between the receptor surface of the olfactory epithelium and the olfactory bulb were discovered. The olfactory bulb is a complexly organized nerve center. There are 6 main layers in the bulb: the layer of olfactory nerve fibers, the glomerular layer, the external synaptic or reticular layer, the layer of mitral neurocytes, and the internal granular layer. Mitral neurocytes serve as the main second-order olfactory neurons. Their axons form projection tracts from the olfactory bulb. The relative sizes of the olfactory bulb vary greatly. In some mammals (macromatics) with a well-developed sense of smell, it can be a third or even half the length of the entire forebrain (Fig. 2). In primates, especially in humans, the olfactory bulb is reduced. The primary projection areas of the olfactory bulb include the anterior olfactory nucleus, prepiriform and periamygdala cortices, olfactory tubercle, and other parts of the forebrain. The primary projection zone of the olfactory bulb, in turn, is connected by afferent and efferent pathways with the centers of the forebrain and diencephalon, including the hippocampus (horn of Ammon), the amygdala, hypothalamus, thalamus, etc.
Traditionally, to the central department of O. a. included the olfactory bulb, the zones of primary and secondary projection of the bulb in the forebrain (ancient cortex, old cortex and subcortical nuclei), united by the term “olfactory brain”. It has been experimentally shown that the functions of most parts of the olfactory brain are associated with the processes of formation of motivations and emotions, memory processes, control of autonomic reactions, etc. (see Limbic system). The olfactory bulb is the only part of the brain whose removal results in complete loss of smell. The areas of primary projection from the olfactory bulb can be considered associative olfactory centers. In these areas, integration of olfactory and other sensory and visceral afferentation occurs. The projections of receptor cells to the olfactory bulb and the projections from the olfactory bulb to other parts of the brain are homolateral. Therefore, with unilateral lesions of various parts of O. a. Olfactory disorders develop on the affected side.
Rice. 3. Schematic representation of the structure and pathways of the olfactory analyzer: 1 - olfactory bulb; 2 - olfactory tract; 3 - olfactory triangle; 4 - intermediate olfactory stripe; 5 - lateral olfactory stripe; 6 - hook; 7 - amygdala; 8 - parahippocampal gyrus; 9 - hippocampal tray; 10 - dentate gyrus; 11 - waist bundle; 12 — arch and commissure of the arch; 13 - plate of the roof of the midbrain; 14 - perforating fibers; 15 - vaulted gyrus; 16 - thalamus; 17 - mastoid-thalamic bundle; 18 - longitudinal medial strip of gray integument; 19 - vault; 20 - olfactory fascicle of the hippocampus; 21 - terminal strip; 22 - medullary strip of the thalamus; 23 - transparent partition; 24 - fimbria of the hippocampus; 25 - anterior commissure; 26 - medial olfactory stripe; 27 - septal-thalamic bundle; 28 — leg of the transparent septum; 29 - olfactory fibers to the gray tubercle; 30 - main olfactory bundle from the mastoid body; 31 - mastoid body; 32 - mastoid-tegmental bundle; 33—bent beam; 34 - interpeduncular node; 35 - cerebral peduncle; 36 - pedicle of the mastoid body; 37 - posterior longitudinal fascicle; 38 - anterior nucleus of the thalamus.
Pathways
- a system of sequentially located neurons forming complex reflex arcs, thanks to which it becomes possible to conduct impulses from the periphery (from olfactory receptor cells) to the cortical and subcortical olfactory centers, and from them to the higher parts of the nervous system (Fig. 3). The first neuron of the olfactory pathway is represented by olfactory cells located in the mucous membrane of the upper nasal passage. The dendrites of these cells end here in the olfactory clubs, and the axons, as part of the olfactory nerve going to the olfactory bulb (bulbus olfactorius), end in mitral neurocytes. This part of the olfactory pathway is called the “peripheral olfactory pathway.” The central olfactory pathway (tractus olfactorius) begins from the mitral neurocytes of the olfactory bulb. The axons of neurons, represented by mitral neurocytes, follow as part of the superficial molecular layer of the olfactory tract towards the olfactory triangle (trigonum olfactorium). Some of the fibers of the olfactory tract are interrupted in the area of accumulation of nerve cells located in the central parts of the olfactory tract, in the olfactory triangle or in the anterior perforated substance (substantia perforata ant.). The axons of neurons, the bodies of which are located in the secondary olfactory cortical centers, are grouped in the form of three olfactory bundles - lateral, intermediate and medial, which are the basis of the olfactory stripes of the same name (striae olfactoriae). The olfactory bundles contain some fibers and mitral neurocytes. The lateral olfactory bundle is the most powerful; it goes directly to the cortical end of the olfactory analyzer - the hook of the parahippocampal gyrus. The intermediate olfactory bundle ends at the cells of the anterior perforated substance of its own and the opposite side, following in the latter case through the anterior commissure. The axons of the cells of the perforated substance pass through the septum pellucidum, the fornix and along the fimbria of the hippocampus rush to the uncus of the parahippocampal gyrus. The internal olfactory bundle, or medial bundle of the forebrain (Wallenberg's main olfactory bundle, radiatio olfactoria basalis;, goes from the olfactory bulb and tract through the deep layers of the anterior perforated substance to the mamillary, or mastoid, bodies (corpora mamillaria). Olfactory-lead bundle (fasc . olfactohabenularis) is represented by fibers traveling as part of the medullary stria of the thalamus (stria medullaris thalami) to the nucleus of the leash (nucleus habenulae), where the descending path associated with the motor neurons of the spinal cord begins. The terminal stria (stria terminalis) lies in the groove between the caudate nucleus and the thalamus (visual thalamus). It contains a bundle of fibers going from the uncus of the parahippocampal gyrus and the amygdala (corpus amygdaloideum) to the anterior perforated substance. The medial olfactory bundle ends at the cells of the subcallosal area (area subcallosa, s. area parolfactoria Broca) and the napa-terminal gyrus (gyrus paraterminalis s. gyrus subcallosus). Axons of cells of the subcallosal field and paraterminal gyrus are directed to the parahippocampal gyrus or to the hippocampus in two ways: above the corpus callosum and below it. Above the corpus callosum, the fibers form two semicircles. Most of them pass in the white matter of the vaulted gyrus (gyrus fornicatus; BNA) as part of the cingulate bundle; the smaller part, forming the second semiring, follows as part of the medial longitudinal stripes of the gray integument (striae longi tu d in ales med.), covering the corpus callosum on top, and then along the ribbon gyrus (gyrus fasciolaris) and the dentate gyrus (gyrus dentatus). Fibers passing under the corpus callosum reach the hippocampus and the uncus of the parahippocampal gyrus, passing through the plate of the transparent septum (lamina septi pellucidi), fornix (fornix) and fimbria hippocampi. There are direct connections between the olfactory center of the parahippocampal gyrus and the hippocampus. The olfactory centers are connected through association pathways to various parts of the cortex, mainly to the hippocampal cortex. In turn, the connection between the hippocampal cortex of the right and left hemispheres and the mammillary bodies is carried out through commissural and projection fibers of the fornix. The mastoid bodies send fibers to various formations of the brain stem, in particular through the main mastoid fascicle (fasc. mamillaris princeps). This fascicle departs from the medial nucleus of the mastoid body and splits into two fascicles: the mastoid-thalamic (fasc. mamillothalamicus), heading to the anterior nucleus of the thalamus, and the mastoid-tegmental (fasc. mamillotegmentalis), following to the tire of the cerebral peduncle, its nuclei, and partly and to the gray matter of the cerebral aqueduct, where the posterior longitudinal fasciculus (fasc. longitudinalis post.) originates, reaching the nuclei of the cranial nerves.
Dysfunction of the olfactory analyzer - see Smell.
Bibliography:
Bogomolova E. M. Olfactory formations of the brain and their biological significance, Usp. fiziol, nauk., vol. 1, no. 4, p. 126, 1970, bibliogr.; Bronstein A.I. Taste and smell (Chemical analyzers of the oral and nasal cavity), M.-L., 1956; Guselnikova K. G. and Guselnikov V. I. Electrophysiology of the olfactory analyzer of vertebrates, M., 1975; Multi-volume guide to neurology, ed. N. I. Grashchenkova, vol. 1, book. 2, p. 194, M., 1957; Sarkisov S. A. Essays on the structure and function of the brain, M., 1964; Physiology of sensory systems, ed. G.V. Gershuni, part 2, p. 515, L., 1972; Adey WR Higher olfactory centres, in the book: Taste a. smell in vertebrates, ed. by GEW Wolstenholme a. J. Knight, p. 357, L., 1970; Clara M. Das Nervensystem des Menschen, Lpz., 1959; Gottschick J. Die Leistungen des Nervensystems, Jena, 1955; Les grandes activites du rhinencephale, publ. par Th. Alajouanine, v. 1-2, P., 1961; Holley A. et MacLeod P. Transduction et codage des informations olfactives chez les vertebres, J. Physiol. (Paris), t. 73, p. 725, 1977, bibliogr.
A. V. Minor; V.V. Turygin (morphology).
Central department
The cells of the olfactory bulb give off processes that form the olfactory tract. The olfactory tract carries impulses to various areas of the brain.
The center of the olfactory analyzer is an area that covers parts of the temporal and frontal lobes of the cerebral cortex. Here the sensation of smell is formed and an adequate reaction to it is given.
Some substances cause other sensations in addition to olfactory ones. The smell of menthol is accompanied by a feeling of cold, formaldehyde - a tingling sensation in the nose, chloroform - a feeling of sweetness.